Tranexamic acid in hemostasis and resuscitation: A comprehensive clinical review
Tranexamic acid in hemostasis and resuscitation
DOI:
https://doi.org/10.54205/ccc.v34.277832Keywords:
Antifibrinolytic agents, Fibrinolysis, Hemorrhage, Hemostasis, Resuscitation, Tranexamic acidAbstract
Tranexamic acid (TXA), a synthetic lysine analogue that stabilizes fibrin clots against premature degradation, has become a cornerstone in the management of acute hemorrhage. Its application has expanded from well-established indications like trauma and postpartum hemorrhage to a variety of other clinical scenarios. However, this widespread use is accompanied by a complex evidence base, with significant variations in dosing, timing, and administration routes, and a growing recognition of conditions where TXA's risk may outweigh its benefit. This narrative review aims to consolidate and critically appraise the evidence from landmark clinical trials and major international guidelines. We explore the established and emerging indications for TXA, delve into its nuanced safety profile, and highlight the critical importance of timely administration. Ultimately, this review provides clinicians with a practical, evidence-based synthesis to guide the safe and effective use of TXA in resuscitation and the management of bleeding.
Downloads
References
Watts G. Utako Okamoto. Lancet. 2016;387:2286.
Relke N, Chornenki NLJ, Sholzberg M. Tranexamic acid evidence and controversies: An illustrated review. Res Pract Thromb Haemost. 2021;5:e12546.
Tengborn L, Blomback M, Berntorp E. Tranexamic acid--an old drug still going strong and making a revival. Thromb Res. 2015;135:231-42.
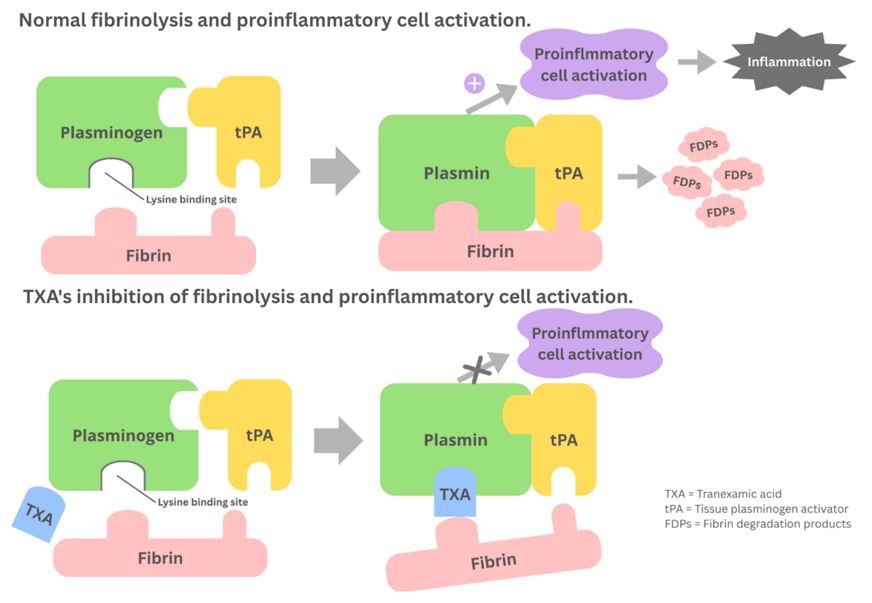
Syrovets T, Lunov O, Simmet T. Plasmin as a proinflammatory cell activator. J Leukoc Biol. 2012;92:509-19.
Ng W, Jerath A, Wasowicz M. Tranexamic acid: a clinical review. Anaesthesiol Intensive Ther. 2015;47:339-50.
Jimenez JJ, Iribarren JL, Lorente L, Rodriguez JM, Hernandez D, Nassar I, et al. Tranexamic acid attenuates inflammatory response in cardiopulmonary bypass surgery through blockade of fibrinolysis: a case control study followed by a randomized double-blind controlled trial. Crit Care. 2007;11:R117.
Draxler DF, Yep K, Hanafi G, Winton A, Daglas M, Ho H, et al. Tranexamic acid modulates the immune response and reduces postsurgical infection rates. Blood Adv. 2019;3:1598-609.
Roberts I, Shakur H, Coats T, Hunt B, Balogun E, Barnetson L, et al. The CRASH-2 trial: a randomised controlled trial and economic evaluation of the effects of tranexamic acid on death, vascular occlusive events and transfusion requirement in bleeding trauma patients. Health Technol Assess. 2013;17:1-79.
Moore HB, Moore EE, Gonzalez E, Chapman MP, Chin TL, Silliman CC, et al. Hyperfibrinolysis, physiologic fibrinolysis, and fibrinolysis shutdown: the spectrum of postinjury fibrinolysis and relevance to antifibrinolytic therapy. J Trauma Acute Care Surg. 2014;77:811-7; discussion 7.
Gayet-Ageron A, Prieto-Merino D, Ker K, Shakur H, Ageron FX, Roberts I, et al. Effect of treatment delay on the effectiveness and safety of antifibrinolytics in acute severe haemorrhage: a meta-analysis of individual patient-level data from 40 138 bleeding patients. Lancet. 2018;391:125-32.
Meizoso JP, Dudaryk R, Mulder MB, Ray JJ, Karcutskie CA, Eidelson SA, et al. Increased risk of fibrinolysis shutdown among severely injured trauma patients receiving tranexamic acid. J Trauma Acute Care Surg. 2018;84:426-32.
Guyette FX, Brown JB, Zenati MS, Early-Young BJ, Adams PW, Eastridge BJ, et al. Tranexamic acid during prehospital transport in patients at risk for hemorrhage after injury: A double-blind, placebo-controlled, randomized clinical trial. JAMA Surg. 2020;156:11-20.
Li SR, Guyette F, Brown J, Zenati M, Reitz KM, Eastridge B, et al. Early prehospital tranexamic acid following injury is associated with a 30-day survival benefit: A secondary analysis of a randomized clinical trial. Ann Surg. 2021;274:419-26.
Investigators PA-T, the ACTG, Gruen RL, Mitra B, Bernard SA, McArthur CJ, et al. Prehospital tranexamic acid for severe trauma. N Engl J Med. 2023;389:127-36.
National Association of Emergency Medical Technicians. PHTLS : Prehospital Trauma Life Support. 10th ed. Burlington (MA): Jones & Bartlett Learning; 2022.
American College of Surgeons Committee on Trauma. Advanced Trauma Life Support (ATLS®) Student Course Manual. 11th ed. Chicago: American College of Surgeons; 2025.
Roberts I, Shakur-Still H, Aeron-Thomas A, Beaumont D, Belli A, Brenner A, et al. Tranexamic acid to reduce head injury death in people with traumatic brain injury: the CRASH-3 international RCT. Health Technol Assess. 2021;25:1-76.
National Institute for Health and Care Excellence. Head injury: assessment and early management [Internet]. London: 2023 [cited 2026 Feb 17]. Available from: https://www.nice.org.uk/guidance/ng232
Sprigg N, Flaherty K, Appleton JP, Al-Shahi Salman R, Bereczki D, Beridze M, et al. Tranexamic acid for hyperacute primary Intracerebral haemorrhage (TICH-2): an international randomised, placebo-controlled, phase 3 superiority trial. Lancet. 2018;391:2107-15.
Meretoja A, Yassi N, Wu TY, Churilov L, Sibolt G, Jeng JS, et al. Tranexamic acid in patients with intracerebral haemorrhage (STOP-AUST): a multicentre, randomised, placebo-controlled, phase 2 trial. Lancet Neurol. 2020;19:980-7.
Liu J, Nie X, Gu H, Zhou Q, Sun H, Tan Y, et al. Tranexamic acid for acute intracerebral haemorrhage growth based on imaging assessment (TRAIGE): a multicentre, randomised, placebo-controlled trial. Stroke Vasc Neurol. 2021;6:160-9.
Polymeris AA, Karwacki GM, Siepen BM, Schaedelin S, Tsakiris DA, Stippich C, et al. Tranexamic acid for Intracerebral hemorrhage in patients on non-vitamin K antagonist oral anticoagulants (TICH-NOAC): a multicenter, randomized, placebo-controlled, phase 2 trial. Stroke. 2023;54:2223-34.
Yassi N, Zhao H, Churilov L, Wu TY, Ma H, Nguyen H-T, et al. Tranexamic acid versus placebo in individuals with intracerebral haemorrhage treated within 2 h of symptom onset (STOP-MSU): an international, double-blind, randomised, phase 2 trial. The Lancet Neurology. 2024;23:577-87.
Steiner T, Purrucker JC, Aguiar de Sousa D, Apostolaki-Hansson T, Beck J, Christensen H, et al. European Stroke Organisation (ESO) and European Association of Neurosurgical Societies (EANS) guideline on stroke due to spontaneous intracerebral haemorrhage. Eur Stroke J. 2025:23969873251340815.
Post R, Germans MR, Tjerkstra MA, Vergouwen MDI, Jellema K, Koot RW, et al. Ultra-early tranexamic acid after subarachnoid haemorrhage (ULTRA): a randomised controlled trial. Lancet. 2021;397:112-8.
Germans MR, Dronkers WJ, Baharoglu MI, Post R, Verbaan D, Rinkel GJ, et al. Antifibrinolytic therapy for aneurysmal subarachnoid haemorrhage. Cochrane Database Syst Rev. 2022;11:CD001245.
Hoh BL, Ko NU, Amin-Hanjani S, Chou S-Y, Cruz-Flores S, Dangayach NS, et al. 2023 Guideline for the management of patients with aneurysmal subarachnoid hemorrhage: A guideline from the american heart association/american stroke association. Stroke. 2023;54:e314-e70.
Treggiari MM, Rabinstein AA, Busl KM, Caylor MM, Citerio G, Deem S, et al. Guidelines for the neurocritical care management of aneurysmal subarachnoid hemorrhage. Neurocrit Care. 2023;39:1-28.
Collaborators WT. Effect of early tranexamic acid administration on mortality, hysterectomy, and other morbidities in women with post-partum haemorrhage (WOMAN): an international, randomised, double-blind, placebo-controlled trial. Lancet. 2017;389:2105-16.
Sentilhes L, Winer N, Azria E, Senat MV, Le Ray C, Vardon D, et al. Tranexamic acid for the prevention of blood loss after vaginal delivery. N Engl J Med. 2018;379:731-42.
Sentilhes L, Madar H, Le Lous M, Senat MV, Winer N, Rozenberg P, et al. Tranexamic acid for the prevention of blood loss after cesarean among women with twins: a secondary analysis of the TRAnexamic Acid for Preventing Postpartum Hemorrhage Following a Cesarean Delivery randomized clinical trial. Am J Obstet Gynecol. 2022;227:889 e1- e17.
Collaborators W-T. The effect of tranexamic acid on postpartum bleeding in women with moderate and severe anaemia (WOMAN-2): an international, randomised, double-blind, placebo-controlled trial. Lancet. 2024;404:1645-56.
Vogel JP, Oladapo OT, Dowswell T, Gulmezoglu AM. Updated WHO recommendation on intravenous tranexamic acid for the treatment of post-partum haemorrhage. Lancet Glob Health. 2018;6:e18-e9.
Lukes AS, Moore KA, Muse KN, Gersten JK, Hecht BR, Edlund M, et al. Tranexamic acid treatment for heavy menstrual bleeding: a randomized controlled trial. Obstet Gynecol. 2010;116:865-75.
Bryant‐Smith AC, Lethaby A, Farquhar C, Hickey M. Antifibrinolytics for heavy menstrual bleeding. Cochrane Database of Systematic Reviews. 2018.
Bofill Rodriguez M, Dias S, Jordan V, Lethaby A, Lensen SF, Wise MR, et al. Interventions for heavy menstrual bleeding; overview of Cochrane reviews and network meta-analysis. Cochrane Database Syst Rev. 2022;5:Cd013180.
Collaborators H-IT. Effects of a high-dose 24-h infusion of tranexamic acid on death and thromboembolic events in patients with acute gastrointestinal bleeding (HALT-IT): an international randomised, double-blind, placebo-controlled trial. Lancet. 2020;395:1927-36.
Gralnek IM, Stanley AJ, Morris AJ, Camus M, Lau J, Lanas A, et al. Endoscopic diagnosis and management of nonvariceal upper gastrointestinal hemorrhage (NVUGIH): European Society of Gastrointestinal Endoscopy (ESGE) Guideline - Update 2021. Endoscopy. 2021;53:300-32.
Triantafyllou K, Gkolfakis P, Gralnek IM, Oakland K, Manes G, Radaelli F, et al. Diagnosis and management of acute lower gastrointestinal bleeding: European Society of Gastrointestinal Endoscopy (ESGE) guideline. Endoscopy. 2021;53:850-68.
Sengupta N, Feuerstein JD, Jairath V, Shergill AK, Strate LL, Wong RJ, et al. Management of patients with acute lower gastrointestinal bleeding: An updated ACG guideline. Am J Gastroenterol. 2023;118:208-31.
Zahed R, Moharamzadeh P, Alizadeharasi S, Ghasemi A, Saeedi M. A new and rapid method for epistaxis treatment using injectable form of tranexamic acid topically: a randomized controlled trial. Am J Emerg Med. 2013;31:1389-92.
Zahed R, Mousavi Jazayeri MH, Naderi A, Naderpour Z, Saeedi M. Topical tranexamic acid compared with anterior nasal packing for treatment of epistaxis in patients taking antiplatelet drugs: Randomized controlled trial. Acad Emerg Med. 2018;25:261-6.
Joseph J, Martinez-Devesa P, Bellorini J, Burton MJ. Tranexamic acid for patients with nasal haemorrhage (epistaxis). Cochrane Database Syst Rev. 2018;12:CD004328.
Reuben A, Appelboam A, Stevens KN, Vickery J, Ewings P, Ingram W, et al. The use of tranexamic acid to reduce the need for nasal packing in epistaxis (NoPAC): Randomized controlled trial. Ann Emerg Med. 2021;77:631-40.
Janapala RN, Tran QK, Patel J, Mehta E, Pourmand A. Efficacy of topical tranexamic acid in epistaxis: A systematic review and meta-analysis. Am J Emerg Med. 2022;51:169-75.
Bellam BL, Dhibar DP, Suri V, Sharma N, Varma SC, Malhotra S, et al. Efficacy of tranexamic acid in haemoptysis: A randomized, controlled pilot study. Pulm Pharmacol Ther. 2016;40:80-3.
Wand O, Guber E, Guber A, Epstein Shochet G, Israeli-Shani L, Shitrit D. Inhaled tranexamic acid for hemoptysis treatment: a randomized controlled trial. Chest. 2018;154:1379-84.
Gopinath B, Mishra PR, Aggarwal P, Nayaka R, Naik SR, Kappagantu V, et al. Nebulized vs IV tranexamic acid for hemoptysis: a pilot randomized controlled trial. Chest. 2023;163:1176-84.
Myles PS, Smith JA, Painter T. Tranexamic acid in patients undergoing coronary-artery surgery. N Engl J Med. 2017;376:1893.
Casselman FPA, Lance MD, Ahmed A, Ascari A, Blanco-Morillo J, Bolliger D, et al. 2024 EACTS/EACTAIC Guidelines on patient blood management in adult cardiac surgery in collaboration with EBCP. J Cardiothorac Vasc Anesth. 2025;39:1964-2018.
Devereaux PJ, Marcucci M, Painter TW, Conen D, Lomivorotov V, Sessler DI, et al. Tranexamic acid in patients undergoing noncardiac surgery. N Engl J Med. 2022;386:1986-97.
National Institute for Health and Care Excellence. Blood transfusion [Internet]. 2016 [cited 2026 Feb 17]. Available from: https://www.nice.org.uk/guidance/ng24
Halvorsen S, Mehilli J, Cassese S, Hall TS, Abdelhamid M, Barbato E, et al. [2022 ESC Guidelines on cardiovascular assessment and management of patients undergoing non-cardiac surgery Developed by the task force for cardiovascular assessment and management of patients undergoing non-cardiac surgery of the European Society of Cardiology (ESC) Endorsed by the European Society of Anaesthesiology and Intensive Care (ESAIC)]. G Ital Cardiol (Rome). 2023;24:e1-e102.
Tsan SEH, Viknaswaran NL, Cheong CC, Cheah S, Ng KT, Mong SXY, et al. Prophylactic intravenous tranexamic acid and thromboembolism in non-cardiac surgery: a systematic review, meta-analysis and trial sequential analysis. Anaesthesia. 2023;78:1153-61.
Park LJ, Marcucci M, Ofori SN, Borges FK, Nenshi R, Kanstrup CTB, et al. Safety and efficacy of tranexamic acid in general surgery. JAMA Surg. 2025;160:267-74.
Delgado LM, Pompeu BF, Martins GHA, Azevedo ML, Pasqualotto E, Chulam TC, et al. Perioperative use of tranexamic acid in general surgery: a systematic review and meta-analysis. JAMA Surg. 2025.
Kumar S, Randhawa MS, Ganesamoni R, Singh SK. Tranexamic acid reduces blood loss during percutaneous nephrolithotomy: a prospective randomized controlled study. J Urol. 2013;189:1757-61.
Bansal A, Arora A. A double-blind, placebo-controlled randomized clinical trial to evaluate the efficacy of tranexamic acid in irrigant solution on blood loss during percutaneous nephrolithotomy: a pilot study from tertiary care center of North India. World J Urol. 2017;35:1233-40.
Cleveland B, Norling B, Wang H, Gandhi V, Price CL, Borofsky M, et al. Tranexamic acid for percutaneous nephrolithotomy: an abridged Cochrane review. BJU Int. 2024;133:259-72.
Kumsar S, Dirim A, Toksoz S, Saglam HS, Adsan O. Tranexamic acid decreases blood loss during transurethral resection of the prostate (TUR -P). Cent European J Urol. 2011;64:156-8.
Tawfick A, Mousa W, El-Zhary AF, Saafan AM. Can tranexamic acid in irrigation fluid reduce blood loss during monopolar transurethral resection of the prostate? A randomised controlled trial. Arab J Urol. 2022;20:94-9.
Tan CW, Chlebicki MP. Urinary tract infections in adults. Singapore Med J. 2016;57:485-90.
Moharamzadeh P, Ojaghihaghighi S, Amjadi M, Rahmani F, Farjamnia A. Effect of tranexamic acid on gross hematuria: A pilot randomized clinical trial study. Am J Emerg Med. 2017;35:1922-5.
Alameel T, West M. Tranexamic Acid treatment of life-threatening hematuria in polycystic kidney disease. Int J Nephrol. 2011;2011:203579.
Maresca G, Royle J, Donaldson JF. Tranexamic acid-induced ureteric clot obstruction in a patient with urothelial cell carcinoma resulting in upper urinary tract perforation. BMJ Case Rep. 2022;15.
Haratian A, Shelby T, Hasan LK, Bolia IK, Weber AE, Petrigliano FA. Utilization of tranexamic acid in surgical orthopaedic practice: Indications and current considerations. Orthop Res Rev. 2021;13:187-99.
Mei L, Li H, Zhu W, Luo Y, Mao X. Efficacy and safety of tranexamic acid in unilateral major revision total hip arthroplasty. Ann Palliat Med. 2020;9:2466-73.
Zhang P, Liang Y, Chen P, Fang Y, He J, Wang J. Combined application versus topical and intravenous application of tranexamic acid following primary total hip arthroplasty: a meta-analysis. BMC Musculoskelet Disord. 2017;18:90.
Liu X, Liu J, Sun G. A comparison of combined intravenous and topical administration of tranexamic acid with intravenous tranexamic acid alone for blood loss reduction after total hip arthroplasty: A meta-analysis. Int J Surg. 2017;41:34-43.
Gernsheimer TB, Brown SP, Triulzi DJ, Key NS, El Kassar N, Herren H, et al. Prophylactic tranexamic acid in patients with hematologic malignancy: a placebo-controlled, randomized clinical trial. Blood. 2022;140:1254-62.
Investigators TT. Tranexamic acid versus placebo to prevent bleeding in patients with haematological malignancies and severe thrombocytopenia (TREATT): a randomised, double-blind, parallel, phase 3 superiority trial. Lancet Haematol. 2025;12:e14-e22.
American Society of Anesthesiologists Task Force on Perioperative Blood M. Practice guidelines for perioperative blood management: an updated report by the American Society of Anesthesiologists Task Force on Perioperative Blood Management*. Anesthesiology. 2015;122:241-75.
Karstensen JG, Ebigbo A, Bhat P, Dinis-Ribeiro M, Gralnek I, Guy C, et al. Endoscopic treatment of variceal upper gastrointestinal bleeding: European Society of Gastrointestinal Endoscopy (ESGE) Cascade Guideline. Endosc Int Open. 2020;8:E990-E7.
Pfizer Medical Information. CYKLOKAPRON® (tranexamic acid) medical information [Internet]. Pfizer medical; 2025 [cited 2026 17 February]. Available from: https://www.pfizermedical.com/cyklokapron
Pfizer Medical Information. Cyklokapron T 500 film-coated tablets and Cyklokapron IV 500 Injection [Internet]. Pfizer medical; 2022 [cited 2026 17 February]. Available from: https://labeling.pfizer.com/ShowLabeling.aspx?id=1135
Imbesi S, Nettis E, Minciullo PL, Di Leo E, Saija A, Vacca A, et al. Hypersensitivity to tranexamic acid: a wide spectrum of adverse reactions. Pharm World Sci. 2010;32:416-9.
Taeuber I, Weibel S, Herrmann E, Neef V, Schlesinger T, Kranke P, et al. association of intravenous tranexamic acid with thromboembolic events and mortality: a systematic review, meta-analysis, and meta-regression. JAMA Surg. 2021;156:e210884.
Murao S, Nakata H, Roberts I, Yamakawa K. Effect of tranexamic acid on thrombotic events and seizures in bleeding patients: a systematic review and meta-analysis. Crit Care. 2021;25:380.
Lecker I, Wang DS, Whissell PD, Avramescu S, Mazer CD, Orser BA. Tranexamic acid-associated seizures: Causes and treatment. Ann Neurol. 2016;79:18-26.
Lin Z, Xiaoyi Z. Tranexamic acid-associated seizures: A meta-analysis. Seizure. 2016;36:70-3.
Kalavrouziotis D, Voisine P, Mohammadi S, Dionne S, Dagenais F. High-dose tranexamic acid is an independent predictor of early seizure after cardiopulmonary bypass. Ann Thorac Surg. 2012;93:148-54.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 The Thai Society of Critical Care Medicine

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.




