The kidney structure and hematology of tank-reared-male slender walking catfish (Clarias nieuhofii Valenciennes, 1840): From the hematopoietic tissue to the peripheral blood cells https://doi.org/10.12982/VIS.2026.071
Main Article Content
Abstract
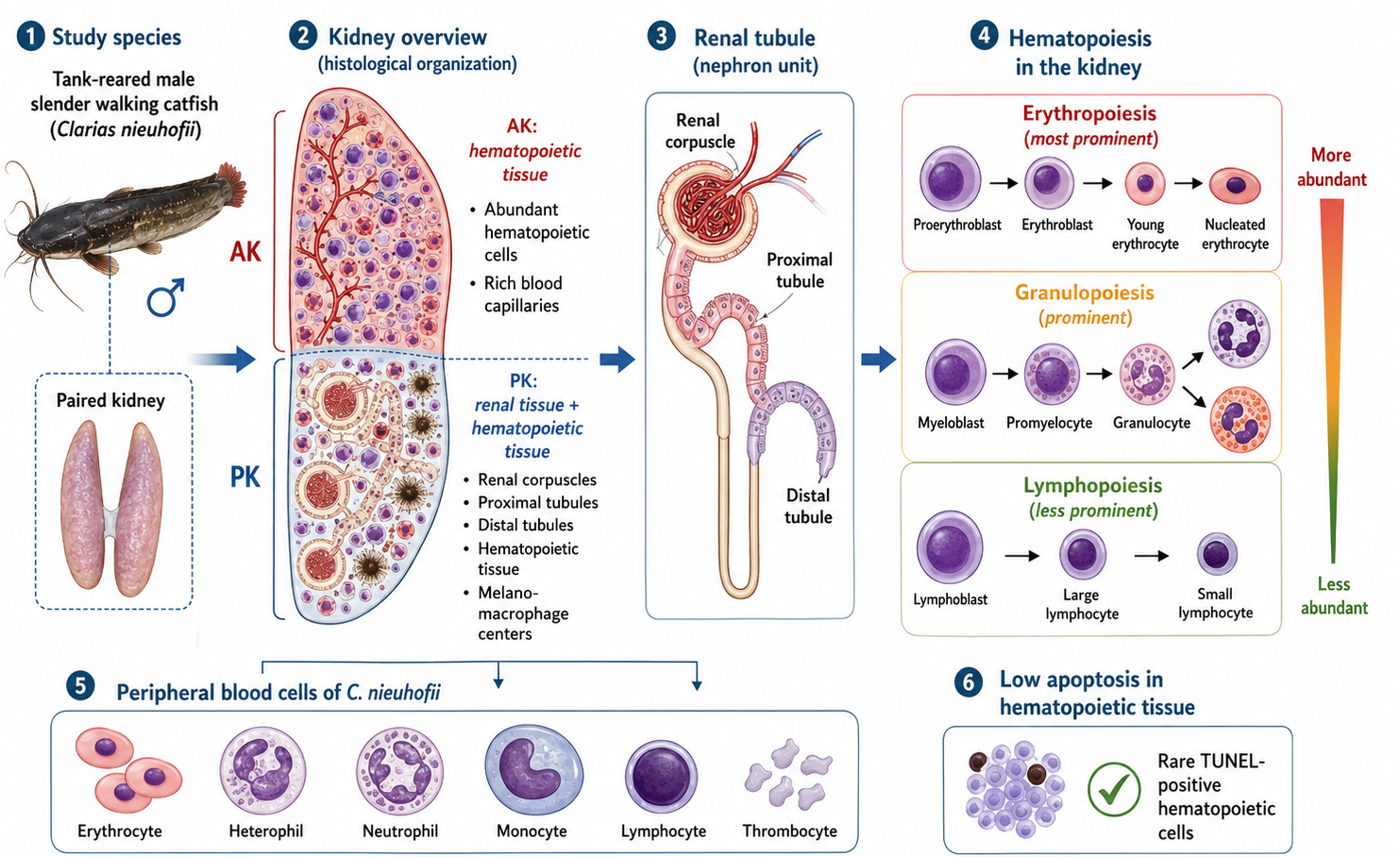
The slender walking catfish Clarias nieuhofii is an economically significant species and a potential candidate for aquaculture in Thailand, but its kidney organization and hematology remain undescribed in the literature. The present study aimed to investigate the hematopoietic structure and ultrastructure of the kidney and peripheral blood cells of male C. nieuhofii obtained from a breeding site in Thailand. Their kidneys were processed for morpho-histology and terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assays using transmission electron microscopy. Blood cell smears were stained with the Alcian Blue/Periodic acid-Schiff method. The kidney of C. nieuhofii was a paired triangular mass comprising anterior (AK) and posterior (PK) regions characterized by renal corpuscle and hematopoietic tissue. In the AK, only hematopoietic tissue and blood capillaries were identified. Hematopoietic tissue was more prominent in the AK than the PK, and its cells were rarely positive in the TUNEL assay. Melano-macrophage centres were observed in the hematopoietic mass in both regions. The detailed hematopoietic ultrastructure was classified into three series, including erythropoiesis, granulopoiesis and lymphopoiesis, which were shared between both kidney regions. Erythropoiesis and granulopoiesis were evident than lymphopoiesis in the kidney tissue. Erythropoietic differentiation was evident. Proerythroblasts were larger than young erythrocytes. Nucleated erythrocytes were observed in peripheral blood smears. The granulopoietic series involved the development of myeloblasts into granulocytes. White blood cells, especially lymphocytes, were also identified. The present study provides useful insights into the renal adaptive histopathology and physiology of C. nieuhofii for aquaculture.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Publishing an article with open access in Veterinary Integrative Sciences leaves the copyright with the author. The article is published under the Creative Commons Attribution License 4.0 (CC-BY 4.0), which allows users to read, copy, distribute and make derivative works from the material, as long as the author of the original work is cited.
References
Abdel-Aziz, E.S.H., Abdu, S.B., Ali, T.E.S., Fouad, H.F., 2010. Haemopoiesis in the head kidney of tilapia, Oreochromis niloticus (Teleostei: Cichlidae): a morphological (optical and ultrastructural) study. Fish Physiol. Biochem. 36, 323–336.
Agius, C., Agbede, S.A., 1984. An electron microscopical study of the genesis of lipofuscin, melanin and hemosiderin in haemopoietic tissues of fish. J. Fish Biol. 24, 471–488.
Agius, C., 1985. The melano-macrophage centers of fish: a review. In: Manning, M.J., Taner, M.F. (Eds.), Fish Immunology. Academic Press, pp. 85–105.
AnvariFar, H., Amirkolaie, A. K., Miandare, H. K., Ouraji, H., Jalali, M. A., & Üçüncü, S. İ., 2017. Apoptosis in fish: environmental factors and programmed cell death. Cell Tissue Res. 368, 425–439.
Bahmani, M., Kazemi, R., Hallagian, A., Sharifpour, E., Mojazi Amiri, B., 2005. Histology of gill, gonad, kidney, liver and digestive system of Persian sturgeon (Acipenser persicus). Iran. J. Fish. Sci. 4(2), 55–66.
Barber, D.L., Westerman, J.E., 1981. The blood cells of the Antarctic icefish Chaenocephalus aceratus Lonnberg: Light and electron microscopic observations. J. Fish Biol. 19, 11–28.
Barber, D.L., Westermann, J.E.M., 1978. Occurrence of the periodic acid-Schiff positive granular leukocyte (PAS-GL) in some fishes and its significance. J. Fish Biol. 12(1), 35–43.
Belosevic, M., Hanington, P.C., Barreda, D.R., 2006. Development of goldfish macrophages in vitro. Fish Shellfish Immunol. 20, 152–171.
Bianchi, M.B., Jeronimo, G.T., Padua, S.B., Satake, F., Ishikawa, M.M., Tavares-Dias M., Martins, M.L., 2014. The haematological profile of farmed Sorubim lima: reference intervals, cell morphology and cytochemistry Vet. Arhiv. 84, 677–690
Blaxhall, P.C., Daisley, K.W., 1973. Routine haematological methods for use with fish blood. J. Fish Biol. 5(6), 771–781.
Boomker, J., 1979. The haemocytology and histology of the haemopoietic organs of Clarias gariepinus and Sarotherodon mossambicus. J. Vet. Res. 46, 217–222.
Borregaard, N., Miller, L.M., 1987. Chemoattractant-regulated mobilization of a novel intracellular compartment in human neutrophils. Science. 237(4819), 1204–1206.
Chen, H., Yuan, G., Su, J., Liu, X., 2019. Hematological analysis of Ctenopharyngodon idella, Megalobrama amblycephala and Pelteobagrus fulvidraco: Morphology, ultrastructure, cytochemistry and quantification of peripheral blood cells. Fish Shellfish Immunol. 90, 376–384.
Chew, X.Z., Gibson-Kueh, S., 2023. The haematology of clinically healthy, farmed juvenile Asian seabass (Lates calcarifer Bloch)—Reference intervals and indicators of subclinical disease. J. Fish Dis. 46(10), 1109–1124.
Clarke, L.R., Cameron, W.A., Carmichael, R.W., 2013. Density effects on subyearling fall Chinook salmon during hatchery rearing in raceways with oxygen supplementation and after release. N. Am. J. Aquac. 75, 18–24.
Dallinger, R., Prosi, F., Segner, H., Back, H., 1987. Contaminated food and uptake of heavy metals by fish: a review and a proposal for further research. Oecologia. 73, 91–98.
Dal’Bó, G.A., Sampaio, F.G., Losekann, M.E., Queiroz, J.F., de Luiz, A.J.B., Wolf, V.H.G., Gonçalves, V.T., Carra, M.L., 2015. Hematological and morphometric blood values of four cultured species of economically important tropical foodfish. Neotrop. Ichthyol. 13(2), 439–446.
Esfandiyari, K., Babaei, M., Morovvati, H., Tarabi, A.M., Kalantari-Hesari, A., 2022. Localization of Na⁺-K⁺-ATPase and Na⁺-K⁺-2Cl⁻ cotransporter, and Na⁺-H⁺ exchanger in the renal system of Walton's mudskipper (Periophthalmus waltoni) using immunohistochemistry and histology methods. Iran. J. Fish. Sci. 21, 1085–1096.
Esteban, M.A., Meseguer, J., Garcia Ayala, A., Agullerio, B., 1989. Erythropoiesis and thrombopoiesis in the head kidney of the sea bass (Dicentrarchus labrax L.): an ultrastructural study. Arch. Histol. Cytol. 52, 407–419.
Fange, R., 1986. Lymphoid organ in sturgeons (Acipenseridae). Vet. Immunol. Immunopathol. 12, 153–161.
Fang, J., Chen, K., Cui, H.M., Peng, X., Tang, L., Zuo, Z.C., 2014. Morphological and cytochemical studies of peripheral blood cells of Schizothorax prenanti. Anat. Histol. Embryol. 43, 386–394.
Feist, S.W., 2009. Atlas of fish histology (Genten, F., Terwinghe, E., Danguy, A., Eds.). J. Fish Biol. 75, 757–758.
Fernandes, C.E., Marcondes, S.F., Galindo, G.M., Franco-Belussi, L., 2019. Kidney anatomy, histology and histometric traits associated to renosomatic index in Gymnotus inaequilabiatus (Gymnotiformes: Gymnotidae). Neotrop Ichthyol. 17, e190107.
Fijan, N., 2002a. Composition of main haematopoietic compartments in normal and bled channel catfish. J. Fish Biol. 60(5), 1142–1154.
Fijan, N., 2002b. Morphogenesis of blood cell lineages in channel catfish. J. Fish Biol. 60(4), 999–1014.
Flerova, E.A., Balabanova, L.V., 2013. Ultrastructure of granulocytes of teleost fish (Salmoniformes, Cypriniformes, Perciformes). J. Evol. Biochem. Physiol. 49(2), 223–233.
Friedrichs, K., Barnhart, K., Blanco, J., Freeman, K., Harr, K., Szladovits, B., Walton, R., 2011. Quality assurance and laboratory standards committee (QALS): Guidelines for the determination of reference intervals in veterinary species and other related topics: Scope. Available online: https://www.asvcp.org/page/QALS_Guidelines.
Gangopadhyay, K., Homechaudhuri, S., 2011. Descriptive characteristics of haemopoietic cell lineages in a facultative air breathing fish Clarias batrachus (L.). Turk. J. Zool. 35(5), 737–746.
Gandahi, N.S., Gandahi, J.A., Yang, P., Tarique, I., Vistro, W.A., Haseeb, A., Chen, Q., 2020. Ultrastructural evidence of melanomacrophagic centers and lipofuscin in the liver of zebrafish (Danio rerio). Zebrafish. 17(2), 83–90.
Gao, Y., Qiang, L., Wu, N., Wang, H., Hao, Y., 2024. Morphological and cytochemical characterization of the peripheral blood cells in Paralichthys olivaceus. Heliyon. 10(17), e37011.
Genovese, K.J., He, H., Lowry, V.K., Swaggerty, C.L., Kogut, M.H., 2006. Comparison of heterophil functions of modern commercial and wild-type Rio Grande turkeys. Avian Pathol. 35, 217–223.
Groman, D.B., 1982. Histology of striped bass. American Fish Society, Maryland.
Groff, J.M., Zinkl, J.G., 1999. Hematology and clinical chemistry of cyprinid fish: Common carp and goldfish. Vet. Clin. North Am. Exot. Anim. Pract. 2(3), 741–746.
Guo, M., Sun, R., Wu, Z., Li, A., Wang, Q., Zhao, Z., Ji, W., 2024. A comparative study on the immune response in the head and trunk kidney of yellow catfish infected with Edwardsiella ictaluri. Fish Shellfish Immunol. 154, 109895.
Harvey, J.W., 2012. Veterinary hematology: a diagnostic guide and color atlas. Elsevier Saunders, St. Louis, Missouri.
Humphrey, R., Bain, J.B., 1990. Endangered animals of Thailand. Sandhill Crane Press, Gainesville, FL.
Imsonpang, S., Kawjaeng, N., Jeamah, A., Senarat, S., Kosiyachinda, P., Kenthao, A., Janyong, S., 2024. Comparative morphology of blood cells of Ambassis nalua, A. vachelli, and Inegocia japonica. Vet. Integr. Sci. 22, 19–27.
Ji, P., Jayapal, S.R., Lodish, H.F., 2008. Enucleation of cultured mouse fetal erythroblasts requires Rac GTPases and mDia2. Nat. Cell Biol. 10, 314–321.
Kent, G.C., Carr, R.K., 2001. Comparative anatomy of the vertebrates, 9th edition. McGraw-Hill, Boston, MA.
Knowles, S., Hrubec, T.C., Smith, S.A., Bakal, R.S., 2006. Hematology and plasma chemistry reference intervals for cultured shortnose sturgeon (Acipenser brevirostrum). Vet. Clin. Pathol. 35(4), 434–440.
Kerr, J.F., Wyllie, A.H., Currie, A.R., 1972. Apoptosis: A basic biological phenomenon with wide-ranging implications in tissue kinetics. Br. J. Cancer. 26(4), 239–257.
Kumar, R., Joy, K.P., Singh, S.M., 2016. Morpho-histology of head kidney of female catfish Heteropneustes fossilis: Seasonal variations in melano-macrophage centers, melanin contents and effects of lipopolysaccharide and dexamethasone on melanins. Fish Physiol. Biochem. 42(5), 1287–1306.
Lawrence, S.M., Corriden, R., Nizet, V., 2018. The ontogeny of a neutrophil: Mechanisms of granulopoiesis and homeostasis. Microbiol. Mol. Biol. Rev. 82(1), e00045-17.
Lertworapreecha, M., Tuyarum, C., Tanomtong, A., Suwannapoom, C., Kiriratnikom, S., 2021. Establishment of cell lines from slender walking catfish Clarias nieuhofii (Valenciennes, 1840). Walailak J. Sci. Technol. 18(12), 10857–10870.
Majeti, R., Park, C.Y., Weissman, I.L., 2007. Identification of a hierarchy of multipotent hematopoietic progenitors in human cord blood. Cell Stem Cell. 1(6), 635–645.
Mattisson, A.G.M., Fänge, R., 1977. Light and electron microscopic observations on the blood cells of the Atlantic hagfish, Myxine glutinosa (L.). Acta Zool. 58(4), 205–221.
Maciak, S., Kostelecka-Myrcha, A., 2011. Regularities of variation of the red blood indices characterizing the respiratory function of blood in selected fish. Zool. Pol. 56(1–4), 35–48.
McCampbell, K.K., Springer, K.N., Wingert, R.A., 2014. Analysis of nephron composition and function in the adult zebrafish kidney. J. Vis. Exp. 90, e51644.
Mesa, M.G., Poe, T.P., Maule, A.G., Schreck, C.B., 1998. Vulnerability to predation and physiological stress responses in juvenile Chinook salmon (Oncorhynchus tshawytscha) experimentally infected with Renibacterium salmoninarum. Can. J. Fish. Aquat. Sci. 55, 1599–1606.
Meseguer, J., Esteban, M.A., Garcia Ayala, A., Lopez Ruiz, A., Agulleiro, B., 1990. Granulopoiesis in the head kidney of the sea bass Dicentrarchus labrax L.: an ultrastructural study. Arch. Histol. Cytol. 53, 287–296.
Megarani, D.V., Hardian, A.B., Arifianto, D., Santosa, C.M., Salasia, S.I., 2020. Comparative morphology and morphometry of blood cells in zebrafish (Danio rerio), common carp (Cyprinus carpio carpio), and tilapia (Oreochromis niloticus). J. Am. Assoc. Lab. Anim. Sci. 59(6), 673–680.
Moradi, A., 2005. Histological structure of kidney in Barbus sharpyei (Doctoral thesis). Faculty of Veterinary Medicine, Shahid Chamran University.
Morovvati, H., Mahabady, M.K., Shahbazi, S., 2012. Histomorphological and anatomical study of kidney in berzem (Barbus pectoralis). Int. J. Fish. Aquac. 4(11), 221–227.
Murtha, J.M., Weici, W., Keller, E.T., 2003. Hematologic and serum biochemical values for the zebrafish (Danio rerio). Comp. Med. 53, 37–41.
Muttakin, I., 2018. Induksi maturasi ikan keli lokal jantan (Clarias nieuhofii) menggunakan hormon GnRH analog + antidopamin (Thesis). Universitas Bangka Belitung.
Nakamura, H., Shimozawa, A., Kikuchi, S.-I., 1993. Melanomacrophage centre-like structure in the heart of the medaka, Oryzias latipes. Ann. Anat. 175, 59–63.
Oh, H.Y., Shin, S.R., Park, J.J., Kim, H.J., Lee, J.S., 2023. Distribution of nephrons in the head kidney of three species of Sebastes (Teleostei: Scorpaenidae). J. Fish Biol. 103(5), 965–973.
Ortiz, M., Esteban, M.Á., 2024. Biology and functions of fish thrombocytes: A review. Fish Shellfish Immunol. 148, 109509.
Palić, D., Beck, L.S., Palić, J., Andreasen, C.B., 2011. Use of rapid cytochemical staining to characterize fish blood granulocytes in species of special concern and determine potential for function testing. Fish Shellfish Immunol. 30(2), 646–652.
Pattnaik, S.T., Samal, S., Mishra, C.S.K., Ghosh, P., Muduli, N., Biswal, M., Sundaray, J.K., 2022. Effect of organophosphate, monocrotophos on behavioural, haematological, histological and biochemical indices in the subtropical catfish Heteropneustes fossilis (Bloch, 1794). J. Indian Soc. Coast. Agric. Res. 40(1), 86–99.
Pechsiri, J., Vanichanon, A., 2016. Genetic diversity in slender walking catfish (Clarias nieuhofii) populations: Implications for population management. Walailak J. Sci. Technol. 13(7), 511–519.
Presnell, J.K., Schreibman, M.P., 2013. Humason’s Animal Tissue Techniques, 5th edition. Johns Hopkins University Press, Baltimore, MD.
Roberts, R.J., Ellis, A.E., 2012. The anatomy and physiology of teleosts. In: Roberts, R.J. (Ed.), Fish Pathology. Wiley-Blackwell, UK, pp. 17–61.
Santos, B.S., Vesagas, F.P.C., Tan, M.T.C., Jumawan, J.C., Quilang, J.P., 2015. Status assessment of Clarias species in the Philippines: Insights from DNA barcodes. Sci. Diliman. 27, 21–40.
Saowakoon, S., Machoo, N., 2021. Chromosome study of slender walking catfish (Clarias nieuhofii) and African catfish (C. gariepinus) F1 hybrid: Revealed by conventional staining and Ag-NOR banding techniques. Food Agric. Sci. Technol. 7(1), 40–48.
Savage, A.G., 1983. The ultrastructure of the blood cells of the pike Esox lucius L. J. Morphol. 178, 187–206.
Schreck, C.B., Jonsson, L., Feist, G., Reno, P., 1995. Conditioning improves performance of juvenile Chinook salmon, Oncorhynchus tswaytscha, to transportation stress. Aquac. 135, 99–110.
Senarat, S., Kettratad, J., Sudtongkong, C., Pengsakul, T., Poolprsert, P., Wongkamhaeng, K., Sukparangsi, W., 2020. Ultrastructure of the granulopoietic cells in head kidney of the Barbour's seahorse, Hippocampus barbouri Jordan & Richardson, 1908 in captivity. J. Appl. Sci. Emerg. Technol. 19(2), 1–10.
Senarat, S., Kettratad, J., Pairohakul, S., Ampawong, S., Huggins, B.P., Coleman, M.M., Kaneko, G., 2022. An update on the evolutionary origin of aglomerular kidney with structural and ultrastructural descriptions of the kidney in three fish species. J. Fish Biol. 100(5), 1283–1298.
Siddiqui, U., Shah, R.H., Rani, A., Tudu, K., Kumar, S., Bisht, H.C.S., Pandey, N., 2020. Comparative study of hematological variation in healthy and fungal infected Kalabans, Bangana dero (Hamilton, 1822). Iran. J. Fish. Sci. 19(1), 501–509.
Shahsavani, D., Mohri, M., 2004. Determination of some blood parameters of fingerling sturgeon (Huso huso) in Guilan province of Iran. J. Appl. Anim. Res. 25(2), 129–130.
Smith, H.M., 1925. Cell size and metabolic activity in amphibia. Biol. Bull. 48, 347–378.
Steinel, N.C., Bolnick, D.I., 2017. Melanomacrophage centers as a histological indicator of immune function in fish and other poikilotherms. Front Immunol. 8, 827.
Sukrakanchana, N., Kiriratnikom, S., Ruangklaii, K., Choksawasdikorn, P., 2012. Haematology of slender walking catfish (Clarias nieuhofii) in culture system. Proceedings of 50th Kasetsart University Annual Conference: Animals, Veterinary Medicine, Fisheries, 534-543
Supriyati, H., Rakhmiyati, R., Luthfi, M.J.F., 2019. Anatomical and histological study of shark (Carcharhinus sorrah) kidney. Biol. Med. Nat. Prod. Chem. 8(2), 37–40.
Suvarna, S.K., Layton, C., Bancroft, J.D., 2013. Bancroft’s Theory and Practice of Histological Techniques. Elsevier, Amsterdam.
Takashima, F., Hibiya, T., 1995. An atlas of fish histology: normal and pathological features. 2nd ed, Tokyo: Kondansha.
Tavares-Dias, M., Barcellos, J.F.M., 2005. Peripheral blood cells of the armoured catfish Hoplosternum littorale Hancock, 1828: A morphological and cytochemical study. Braz. J. Morphol. Sci. 22(4), 215–220.
Tooze, J., Davies, H.G., 1976. Light and electron microscope studies on the spleen of the newt Triturus cristalus: The fine structure of erythropoietic cells. J. Cell Sci. 2, 617–640.
Tsujii, T., Seno, S., 1990. Melano-macrophage centers in the aglomerular kidney of the seahorse (Teleosts): Morphologic studies on its formation and possible function. Anat. Rec. 226, 460–470.
Ueda, I.K., Egami, M.I., Sasso, W., Matushima, E.R., 2001. Cytochemical aspects of the peripheral blood cells of Oreochromis niloticus (Linnaeus, 1758) (Cichlidae, Teleostei): Part II. Braz. J. Vet. Res. Anim. Sci. 38(6), 273–277.
Vidthayanon, C., 2005. Thailand Red Data: Fishes. Office of Natural Resources and Environmental Policy and Planning, Bangkok.
Venkatachalam, U., Venkatachalam, R., Ganesh, K., Aathi, K., 2012. Induction of triploidy catfish through cold shock and heat shock in Clarias batrachus species. Int. J. Aquac. Sci. 2(1), 63–72.
Val, A., 2000. Organic phosphates in the red blood cells of fish. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 125(4), 417–435.
Wang, X., Wu, Z., Wu, S., Chen, X., Hanif, M., Zhang, S., 2021. Hematological and cytochemical characteristics of peripheral blood cells in the argus snakehead (Ophiocephalus argus Cantor). PeerJ. 9, e11234.
Watson, C.F., Baer, K.N., Benson, W.H., 1989. Dorsal gill incision: A simple method for obtaining blood samples in small fish. Environ. Toxicol. Chem. 8, 457–461.
Wilson, J.M., Bunte, R.M., Carty, A.J., 2009. Evaluation of rapid cooling and tricaine methanesulfonate (MS 222) as methods of euthanasia in zebra fish (Danio rerio). J. Am. Assoc. Lab. Anim. Sci. 48, 785–789.
Witeska, M., 2013. Erythrocytes in teleost fishes: A review. Zool. Ecol. 23 (4), 275–281.
Witeska, M., Kondera, E., Ługowska, K., Bojarski, B., 2022. Hematological methods in fish – Not only for beginners. Aquaculture. 547, 737498.
Witeska, M., Kondera, E., Bojarski, B., 2023. Hematological and hematopoietic analysis in fish toxicology — A review. Animals. 13(16), 2625.
Willett, C., Cortes, A., Zuasti, A., Zapata, A., 1999. Early hematopoiesis and developing lymphoid organs in zebrafish. Dev. Dyn. 214, 323–336.
Wyllie, A.H., 1987. Apoptosis: Cell death in tissue regulation. J. Pathol. 153(4), 313–316.
Yamamoto, M., Iuchi, I., 1976. Electron microscopic study of erythrocytes in developing rainbow trout Salmo gairdneri with particular reference to changes in the cell line. J. Exp. Zool. 191(3), 407–426.
Yilmaz, M., Guven, O., Mutaf, B.F., 2015. Comparative morphometry of erythrocytes of different fish species. J. Mol. Cell Biol. 1, 002.
Zapata, A.G., Chibá, A., Varas, A., 1996. Cells and tissues of the immune system of fish. In: Iwama, G., Nakanishi, T. (Eds.), The fish immune system: organism, pathogen, and environment. Academic Press, pp. 1–53.
Zhong, A., Yan, X., 2025. Erythropoiesis in teleost fishes: The fantastic biological process. Rev. Aquac. 17(1), e12960.
Zuasti, A., Ferrer, C., 1988. Granulopoiesis in the head kidney of Sparus auratus. Arch. Histol. Cytol. 51, 425–431.
Zuasti, A., Ferrer, C., 1989. Haemopoiesis in the head kidney of Sparus auratus. Arch. Histol. Cytol. 52(3), 249–255.