Comparative Study of Immunogenicity and reactogenicity of ChAdOx1 vaccine in patients with autoimmune inflammatory rheumatic diseases and healthy controls
คำสำคัญ:
COVID-19, SARS-CoV-2 vaccine, Immunogenicity, Reactogenicity, Autoimmune inflammatory rheumatic diseases (AIRDs)บทคัดย่อ
The data are limited on vaccine responses in patients with autoimmune inflammatory rheumatic disease (AIRD), especially for the adenoviral vector ChAdOx1 vaccine. This single-center prospective cohort study evaluated the immunogenicity and safety of ChAdOx1 vaccine in patients with AIRD at Chulabhorn Hospital, Thailand. Thirty-five patients with AIRDs were enrolled between June 9 and July 1, 2021. The immune response and reactogenicity data of 70 healthcare workers (age- and sex-frequency matched) were used as a control group. All participants were vaccinated with two doses of ChAdOx1 vaccine with a 3-month interval. Antibodies against the receptor-binding domain of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spike protein were assessed prevaccination, 3 months after the first dose, and 1 month after the second dose. Disease activity and reactogenicity were assessed before and 7 days after both vaccinations.
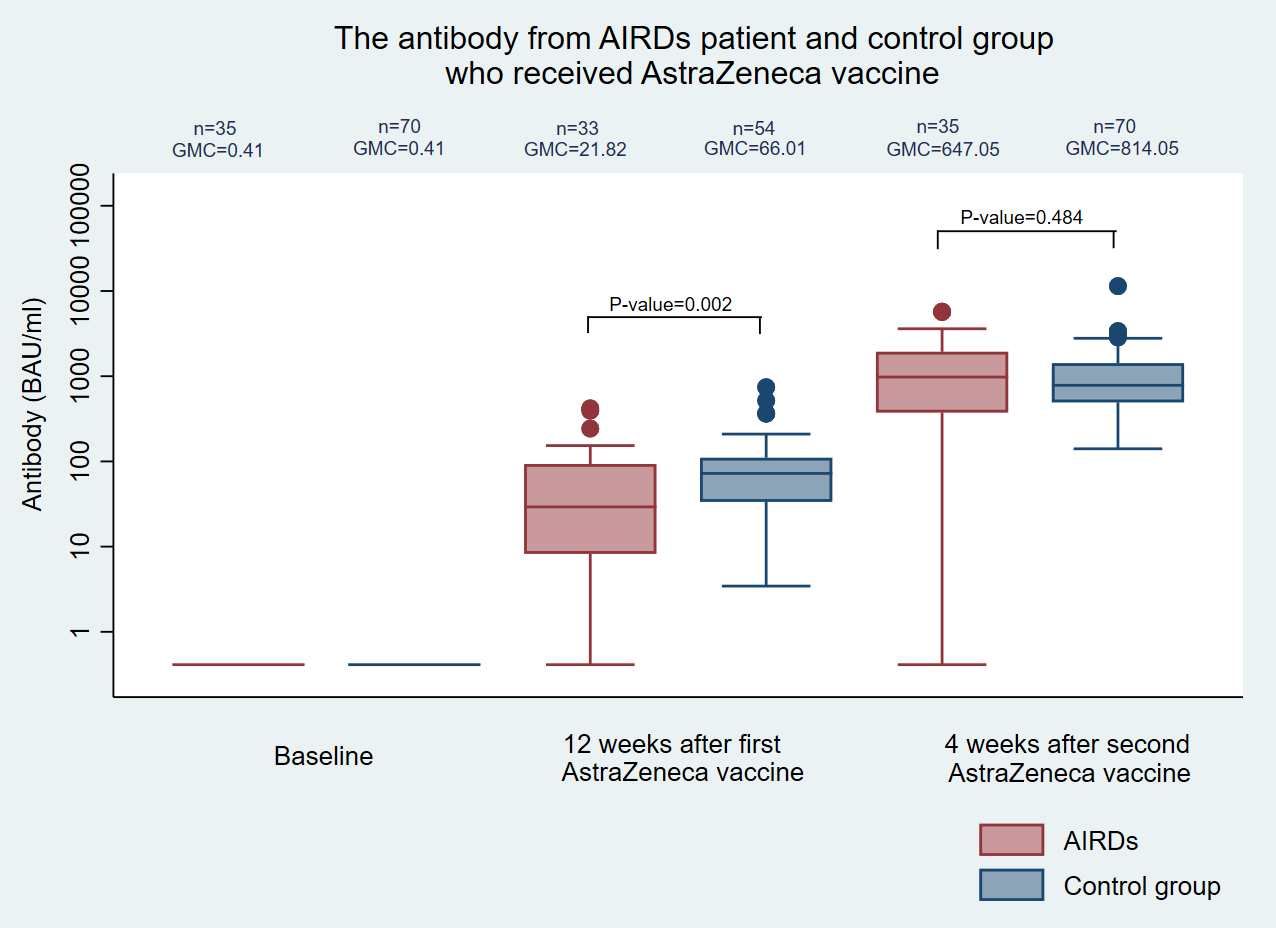
The geometric mean concentration of SARS-CoV-2 antibodies at 3 months after the first vaccination was significantly lower in patients than that in healthy controls (21.82 vs 66.01 BAU/mL, P = 0.002), although there was an insignificant decrease in antibody response in patients with AIRDs compared with that in healthy controls at 1 month after the second vaccination (647.05 vs 814.08 BAU/mL, P = 0.484). Vaccines were mostly well tolerated with mild adverse reactions, and no patients experienced a disease flare. The antibody responses of patients with AIRDs did not significantly differ from those of healthy controls but did tend to be lower. Most vaccinations were well-tolerated without postvaccination disease flares.
Downloads
เอกสารอ้างอิง
Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054-1062. doi:10.1016/S0140-6736(20)30566-3
Kim MJ, Lee EB, Song YW, Park JK. Profile of common inflammatory markers in treatment-naïve patients with systemic rheumatic diseases. Clin Rheumatol. 2020;39(10):2899-2906. doi:10.1007/s10067-020-05049-9
Hyrich KL, Machado PM. Rheumatic disease and COVID-19: epidemiology and outcomes. Nat Rev Rheumatol. 2021;17(2):71-72. doi:10.1038/s41584-020-00562-2
Akiyama S, Hamdeh S, Micic D, Sakuraba A. Prevalence and clinical outcomes of COVID-19 in patients with autoimmune diseases: a systematic review and meta-analysis. Ann Rheum Dis. 2021;80(3):384-391. doi:10.1136/annrheumdis-2020-218946
Listing J, Gerhold K, Zink A. The risk of infections associated with rheumatoid arthritis, with its comorbidity and treatment. Rheumatology (Oxford). 2013;52(1):53-61. doi:10.1093/rheumatology/kes305
Doran MF, Crowson CS, Pond GR, O'Fallon WM, Gabriel SE. Frequency of infection in patients with rheumatoid arthritis compared with controls: a population-based study. Arthritis Rheum. 2002;46(9):2287-2293. doi:10.1002/art.10524
Hasseli R, Mueller-Ladner U, Hoyer BF, et al. Older age, comorbidity, glucocorticoid use and disease activity are risk factors for COVID-19 hospitalisation in patients with inflammatory rheumatic and musculoskeletal diseases. RMD Open. 2021;7(1):e001464. doi:10.1136/rmdopen-2020-001464
Gianfrancesco M, Hyrich KL, Al-Adely S, et al. Characteristics associated with hospitalisation for COVID-19 in people with rheumatic disease: data from the COVID-19 Global Rheumatology Alliance physician-reported registry. Ann Rheum Dis. 2020;79(7):859-866. doi:10.1136/annrheumdis-2020-217871
Voysey M, Clemens SAC, Madhi SA, et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet. 2021;397(10269):99-111. doi:10.1016/S0140-6736(20)32661-1
Liao HT, Tung HY, Chou CT, Tsai HC, Yen YN, Tsai CY. Immunogenicity of the mRNA-1273 and ChAdOx1 nCoV-19 vaccines in Asian patients with autoimmune rheumatic diseases under biologic and/or conventional immunosuppressant treatments. Scand J Rheumatol. 2022;51(6):500-505. doi:10.1080/03009742.2022.2062822
Geisen UM, Berner DK, Tran F, et al. Immunogenicity and safety of anti-SARS-CoV-2 mRNA vaccines in patients with chronic inflammatory conditions and immunosuppressive therapy in a monocentric cohort. Ann Rheum Dis. 2021;80(10):1306-1311. doi:10.1136/annrheumdis-2021-220272
Boyarsky BJ, Ruddy JA, Connolly CM, et al. Antibody response to a single dose of SARS-CoV-2 mRNA vaccine in patients with rheumatic and musculoskeletal diseases. Ann Rheum Dis. 2021;80(8):1098-1099. doi:10.1136/annrheumdis-2021-220289
Furer V, Eviatar T, Zisman D, et al. Immunogenicity and safety of the BNT162b2 mRNA COVID-19 vaccine in adult patients with autoimmune inflammatory rheumatic diseases and in the general population: a multicentre study. Ann Rheum Dis. 2021;80(10):1330-1338. doi:10.1136/annrheumdis-2021-220647
Simon D, Tascilar K, Fagni F, et al. SARS-CoV-2 vaccination responses in untreated, conventionally treated and anticytokine-treated patients with immune-mediated inflammatory diseases. Ann Rheum Dis. 2021;80(10):1312-1316. doi:10.1136/annrheumdis-2021-220461
Shenoy P, Ahmed S, Cherian S, et al. Immunogenicity of the ChAdOx1 nCoV-19 and the BBV152 vaccines in patients with autoimmune rheumatic diseases. bioRxiv. Published online 2021. doi:10.1101/2021.06.06.21258417
Winthrop KL, Bingham CO 3rd, Komocsar WJ, et al. Evaluation of pneumococcal and tetanus vaccine responses in patients with rheumatoid arthritis receiving baricitinib: results from a long-term extension trial substudy. Arthritis Res Ther. 2019;21(1):102. Published 2019 Apr 18. doi:10.1186/s13075-019-1883-1
Day AL, Winthrop KL, Curtis JR. The effect of disease-modifying antirheumatic drugs on vaccine immunogenicity in adults. Cleve Clin J Med. 2020;87(11):695-703. Published 2020 Nov 2. doi:10.3949/ccjm.87a.20056
Teijaro JR, Farber DL. COVID-19 vaccines: modes of immune activation and future challenges. Nat Rev Immunol. 2021;21(4):195-197. doi:10.1038/s41577-021-00526-x
Papp KA, Haraoui B, Kumar D, et al. Vaccination Guidelines for Patients With Immune-Mediated Disorders on Immunosuppressive Therapies. J Cutan Med Surg. 2019;23(1):50-74. doi:10.1177/1203475418811335
Friedman MA, Winthrop KL. Vaccines and Disease-Modifying Antirheumatic Drugs: Practical Implications for the Rheumatologist. Rheum Dis Clin North Am. 2017;43(1):1-13. doi:10.1016/j.rdc.2016.09.003
Tangkum P, Kasitanon N, Gumtorntip W, et al. COVID-19 Vaccination in Patients With Systemic Lupus Erythematosus: Adverse Events and Rating Agreement of Flares Between Patients and Physicians. Int J Rheum Dis. 2024;27(12):e70001. doi:10.1111/1756-185X.70001
ดาวน์โหลด
เผยแพร่แล้ว
รูปแบบการอ้างอิง
ฉบับ
ประเภทบทความ
สัญญาอนุญาต
ลิขสิทธิ์ (c) 2025 ราชวิทยาลัยจุฬาภรณ์

อนุญาตภายใต้เงื่อนไข Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
บทความที่ได้รับการตีพิมพ์เป็นลิขสิทธิ์ของราชวิทยาลัยจุฬาภรณ์
ข้อความที่ปรากฏในบทความแต่ละเรื่องในวารสารวิชาการเล่มนี้เป็นความคิดเห็นส่วนตัวของผู้เขียนแต่ละท่านไม่เกี่ยวข้องกับราชวิทยาลัยจุฬาภรณ์ และคณาจารย์ท่านอื่น ในราชวิทยาลัยฯ แต่อย่างใด ความรับผิดชอบองค์ประกอบทั้งหมดของบทความแต่ละเรื่องเป็นของผู้เขียนแต่ละท่าน หากมีความผิดพลาดใด ๆ ผู้เขียนแต่ละท่านจะรับผิดชอบบทความของตนเองแต่ผู้เดียว


