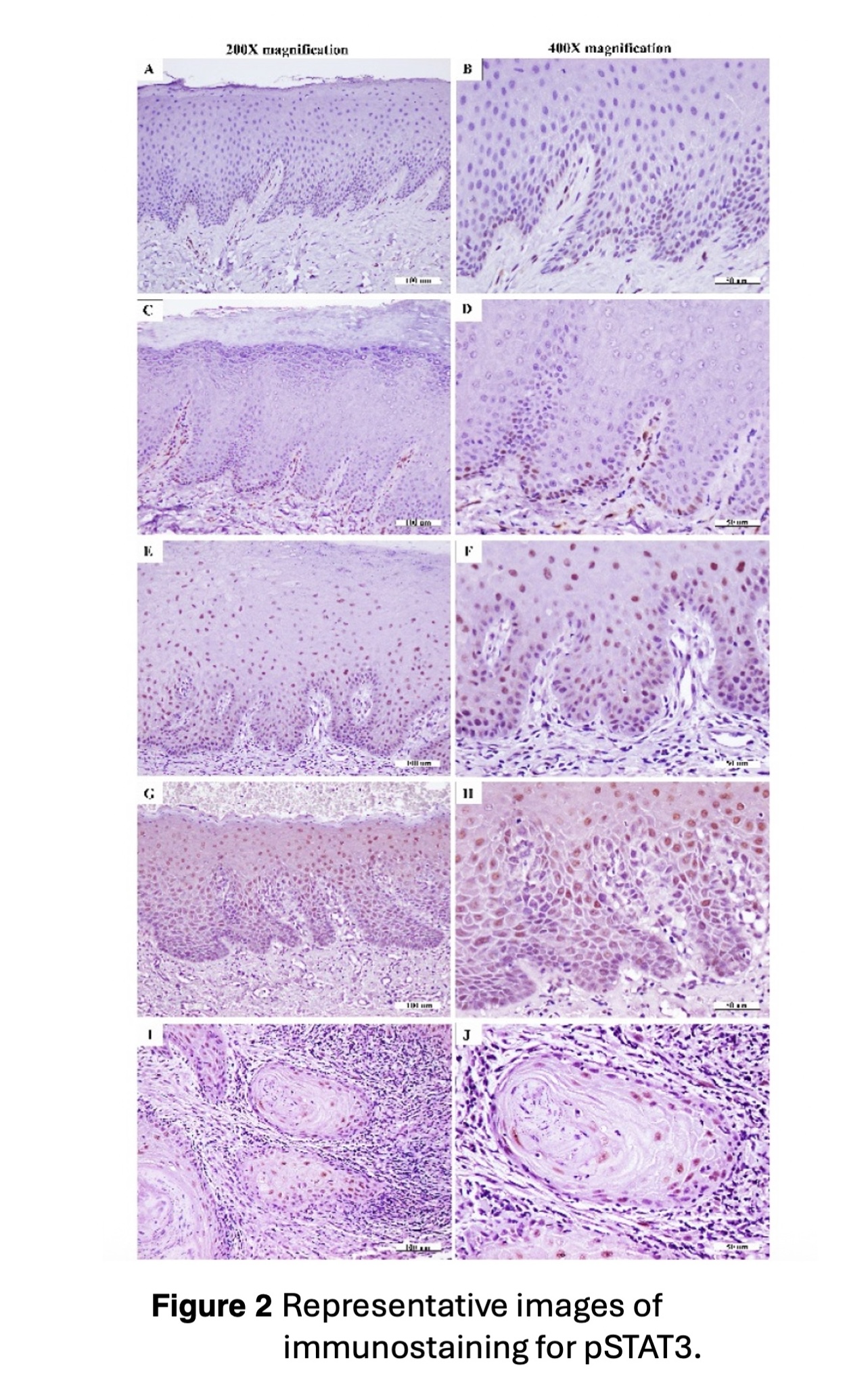
Dynamic transition of pSTAT3 expression demonstrates the highest level in severe oral epithelial dysplasia before downregulation in invasive carcinoma
Main Article Content
Abstract
ThailandThis study aimed to preliminarily evaluate the expression of phosphorylated signal transducer and activator of transcription 3 (pSTAT3) in oral epithelial dysplasia (OED) and oral squamous cell carcinoma (OSCC) in comparison with normal oral mucosa (NOM). Additionally, its potential involvement in the sequential process of oral carcinogenesis was investigated. This study included 75 formalin-fixed, paraffin-embedded tissue specimens, comprising 15 cases each of NOM, mild OED, moderate OED, severe OED, and OSCC. Immunohistochemical analysis was performed to evaluate protein expression. The proportion of positive cells and the staining intensity were semi-quantitatively assessed and recorded as an immunoreactive score (IRS). Comparisons of IRS were conducted among the NOM group, the various grades of OED, and the OSCC group using appropriate statistical analyses. pSTAT3 was found predominantly in the nuclei of epithelial cells, with mild cytoplasmic staining observed to a lesser extent. Immunostaining demonstrated a progressive expansion of positivity from the basal layers in NOM to the superficial epithelial layers in severe OED. Based on the IRS, pSTAT3 expression gradually increased from NOM through the OED groups and declined in the OSCC group. The highest IRS was observed in severe OED compared with NOM (p = 0.002). However, pSTAT3 levels significantly decreased in OSCC compared with severe OED (p = 0.007). pSTAT3 expression progressively increases from NOM to OED, with expansion from basal to full epithelial thickness as the severity of dysplasia increases. These results imply a role of pSTAT3 in early oral carcinogenesis.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
References
Sarode G, Maniyar N, Sarode SC, Jafer M, Patil S, Awan KH. Epidemiologic aspects of oral cancer. Dis Mon. 2020 Dec;66(12):100988. doi: 10.1016/j.disamonth.2020.100988.
Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024 May-Jun;74(3):229-263. doi: 10.3322/caac.21834.
Warnakulasuriya S, Kujan O, Aguirre-Urizar JM, Bagan JV, González-Moles MÁ, Kerr AR, et al. Oral potentially malignant disorders: A consensus report from an international seminar on nomenclature and classification, convened by the WHO Collaborating Centre for Oral Cancer. Oral Dis. 2021 Nov;27(8):1862-1880. doi: 10.1111/odi.13704.
Speight PM, Khurram SA, Kujan O. Oral potentially malignant disorders: risk of progression to malignancy. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018 Jun;125(6):612-627. doi: 10.1016/j.oooo.2017.12.011.
Rogers SN, Lowe D. Health-related quality of life after oral cancer treatment: 10-year outcomes. Oral Surg Oral Med Oral Pathol Oral Radiol. 2020 Aug;130(2):144-149. doi: 10.1016/j.oooo.2020.02.018.
Cabral LGS, Martins IM, Paulo EPA, Pomini KT, Poyet JL, Maria DA. Molecular mechanisms in the carcinogenesis of oral squamous cell carcinoma: A Literature Review. Biomolecules. 2025 Apr;15(5):621. doi: 10.3390/biom15050621.
Crawford M, Liu X, Cheng YL, Tsai RY. Nucleostemin upregulation and STAT3 activation as early events in oral epithelial dysplasia progression to squamous cell carcinoma. Neoplasia. 2021 Dec;23(12):1289-1299. doi: 10.1016/j.neo.2021.11.001.
Geiger JL, Grandis JR, Bauman JE. The STAT3 pathway as a therapeutic target in head and neck cancer: Barriers and innovations. Oral Oncol. 2016 May;56:84-92. doi: 10.1016/j.oraloncology.2015.11.022.
Tashiro K, Oikawa M, Miki Y, Takahashi T, Kumamoto H. Immunohistochemical assessment of growth factor signaling molecules: MAPK, Akt, and STAT3 pathways in oral epithelial precursor lesions and squamous cell carcinoma. Odontology. 2020 Jan;108(1):91-101. doi: 10.1007/s10266-019-00428-4.
Suryawanshi H, Hande A, Aileni KR, Sahu M, Patil SR, Karobari MI. Exploring diagnostic frontiers in oral squamous cell carcinoma: A comprehensive review from immunohistochemistry to genomic profiling. Oral Oncology Reports. 2024 Sep;11:100554. doi: 10.1016/j.oor.2024.100554.
Almangush A, Leivo I, Mäkitie AA. Biomarkers for immunotherapy of oral squamous cell carcinoma: Current status and challenges. Front Oncol. 2021 Mar;11:616629. doi: 10.3389/fonc.2021.616629.
Kaemmerer D, Peter L, Lupp A, Schulz S, Sänger J, Baum RP, et al. Comparing of IRS and Her2 as immunohistochemical scoring schemes in gastroenteropancreatic neuroendocrine tumors. Int J Clin Exp Pathol. 2012 ;5(3):187-194. https://pubmed.ncbi.nlm.nih.gov/22558472/
Monteiro MCdLJ, Tucci R, Oliveira JCd, Brown AL, Araujo NSd, Araujo VCd, et al. Signal transducer and activator of transcription expression 3 (STAT3) in potentially malignant oral lesions. J Oral Diagn. 2019 Jan;4:1-6. doi: 10.5935/2525-5711.20190024.
Fasanaro E, Staffieri C, Cappellesso R, Marino F, Ottaviano G, Val M, et al. Prognostic significance of serine-phosphorylated STAT3 expression in pT1-T2 oral tongue carcinoma. Clin Exp Otorhinolaryngol. 2015 Sep;8(3):275-280. doi: 10.3342/ceo.2015.8.3.275.
Cury PR, Furuse C, de Araújo NS, de Araújo VC. Signal transducer and activator of transcription-3 expression and activation is dysregulated in actinic cheilitis. J Cutan Pathol. 2007 Aug;34(8):606-611. doi: 10.1111/j.1600-0560.2006.00668.x.
Wu M, Song D, Li H, Yang Y, Ma X, Deng S, et al. Negative regulators of STAT3 signaling pathway in cancers. Cancer Manag Res. 2019 May;11:4957-4969. doi: 10.2147/CMAR.S206175.
Alexander WS, Hilton DJ. The role of suppressors of cytokine signaling (SOCS) proteins in regulation of the immune response. Annu Rev Immunol. 2004 ;22:503-529. doi: 10.1146/annurev.immunol.22.091003.090312.
Yoshimura A, Naka T, Kubo M. SOCS proteins, cytokine signalling and immune regulation. Nat Rev Immunol. 2007 Jun;7(6):454-65. doi: 10.1038/nri2093.
Tan Y, Wang Z, Xu M, Li B, Huang Z, Qin S, et al. Oral squamous cell carcinomas: state of the field and emerging directions. Int J Oral Sci. 2023 Sep;15(1):44. doi: 10.1038/s41368-023-00249-w.
Wang Y, Wang Q, Tang CH, Chen HD, Hu GN, Shao JK, et al. p-STAT3 expression in breast cancer correlates negatively with tumor size and HER2 status. Medicine (Baltimore). 2021 Mar;100(10):e25124. doi: 10.1097/MD.0000000000025124.
Walch-Rückheim B, Pahne-Zeppenfeld J, Fischbach J, Wickenhauser C, Horn LC, Tharun L, et al. STAT3/IRF1 pathway activation sensitizes cervical cancer cells to chemotherapeutic drugs. Cancer Res. 2016 Jul;76(13):3872-3883. doi: 10.1158/0008-5472.CAN-14-1306.
Hashimoto S, Hashimoto A, Muromoto R, Kitai Y, Oritani K, Matsuda T. Central roles of STAT3-mediated signals in onset and development of cancers: Tumorigenesis and Immunosurveillance. Cells. 2022 Aug;11(16):2618. doi: 10.3390/cells11162618.
Szczepanek K, Chen Q, Derecka M, Salloum FN, Zhang Q, Szelag M, et al. Mitochondrial-targeted signal transducer and activator of transcription 3 (STAT3) protects against ischemia-induced changes in the electron transport chain and the generation of reactive oxygen species. J Biol Chem. 2011 Aug;286(34):29610-29620. doi: 10.1074/jbc.M111.226209.
Zhang Q, Raje V, Yakovlev VA, Yacoub A, Szczepanek K, Meier J, et al. Mitochondrial localized Stat3 promotes breast cancer growth via phosphorylation of serine 727. J Biol Chem. 2013 Oct;288(43):31280-31288. doi: 10.1074/jbc.M113.505057.
Li Q, Tie Y, Alu A, Ma X, Shi H. Targeted therapy for head and neck cancer: signaling pathways and clinical studies. Signal Transduct Target Ther. 2023 Jan;8(1):31. doi: 10.1038/s41392-022-01297-0.
Qureshy Z, Li H, Zeng Y, Rivera J, Cheng N, Peterson CN, et al. STAT3 Activation as a predictive biomarker for ruxolitinib response in head and neck cancer. Clin Cancer Res. 2022 Nov;28(21):4737-4746. doi: 10.1158/1078-0432.CCR-22-0744.
Picon H, Guddati AK. Mechanisms of resistance in head and neck cancer. Am J Cancer Res. 2020 Sep;10(9):2742-2751. https://pmc.ncbi.nlm.nih.gov/articles/PMC7539768/
Wong ALA, Hirpara JL, Pervaiz S, Eu JQ, Sethi G, Goh BC. Do STAT3 inhibitors have potential in the future for cancer therapy? Expert Opin Investig Drugs. 2017 Aug;26(8):883-887. doi: 10.1080/13543784.2017.1351941.
Tsimberidou AM, Vining DJ, Arora SP, de Achaval S, Larson J, Kauh J, et al. Phase I trial of TTI-101, a first-in-class oral inhibitor of STAT3, in patients with advanced solid tumors. Clin Cancer Res. 2025 Mar;31(6):965-974. doi: 10.1158/1078-0432.CCR-24-2920.
Sen M, Joyce S, Panahandeh M, Li C, Thomas SM, Maxwell J, et al. Targeting Stat3 abrogates EGFR inhibitor resistance in cancer. Clin Cancer Res. 2012 Sep;18(18):4986-4996. doi: 10.1158/1078-0432.CCR-12-0792.