Long Term Outcomes and Durability of Bioprosthetic Valve for Valve Replacement at Siriraj Hospital
DOI:
https://doi.org/10.33192/Smj.2022.26Keywords:
Tissue valve, bioprosthesis valve, structural valve deteriorationAbstract
Objective: Bioprosthesis has been used in cardiac valve replacement for a long time. However, structural valve deterioration is still a major cause of failure. There are several risk factors for valve deterioration. This study evaluates the risk factors of valve deterioration in the long term (10 years) at Siriraj Hospital.
Materials and Methods: We retrospectively reviewed the medical records of 249 patients who underwent mitral or aortic valve replacement between January 2006 and December 2012 using various tissue valves, comprising Carpentier–Edwards porcine, Carpentier–Edwards Perimount bovine pericardial, Carpentier–Edwards Perimount Magna bovine pericardial, and St Jude Trifecta bovine pericardial types. The information from each patient was entered into a database at the time of the operation and followed up regularly, with a mean follow-up of 10 years.
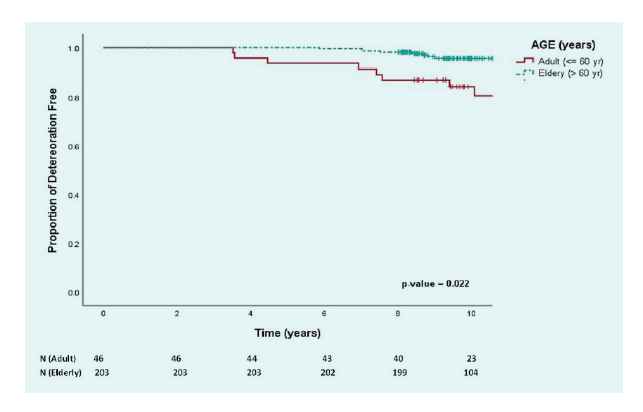
Results: After 10 years follow-up time, the incidence of valve deterioration events were 1.2% and 8.43% in the first five and ten years, respectively. The overall death rate during follow-up was 2.41%. There were three statistically significant risk factors (p < 0.05) of valve deterioration: gender (female) (p = 0.042), age ≤ 60 years old (p = 0.010) and St Jude Trifecta bovine pericardial valve (p = 0.004).
Conclusion: In the surgical populations who underwent valve replacement at Siriraj Hospital with tissue valves, we found an acceptable long-term durability of the tissue valve. The risk factors of valve deterioration were female gender, age ≤ 60 years old, and St Jude Trifecta bovine pericardial valve.
References
Carpentier A, Lemaigre G, Robert L, Carpentier S, Dubost C. Biological factors affecting long-term results of valvular heterografts. J Thorac Cardiovasc Surg. 1969;58(4):467-83.
Russo M, Taramasso M, Guidotti A, Pozzoli A, Segesser L, Nietlispach F, et al. The evolution of surgical valves. Cardiovascular Medicine. 2017;20(12):285-92.
Rizzoli G, Bottio T, Thiene G, Toscano G, Casarotto D. Longterm durability of the Hancock II porcine bioprosthesis. J Thorac Cardiovasc Surg. 2003;126(1):66-74.
O’Gara PT. Chapter 23: Prosthetic Heart Valves. In: Catherine Otto RB, Catherine Otto, Robert Bonow, editor. Valvular Heart Disease: A Companion to Braunwald’s Heart Disease. 3rd ed: Elsevier; 2009. p. 383-98.
Gao G, Wu Y, Grunkemeier GL, Furnary AP, Starr A. Durability of pericardial versus porcine aortic valves. J Am Coll Cardiol. 2004;44(2):384-8.
Hammermeister K, Sethi GK, Henderson WG, Grover FL, Oprian C, Rahimtoola SH. Outcomes 15 years after valve replacement with a mechanical versus a bioprosthetic valve: final report of the Veterans Affairs randomized trial. J Am Coll Cardiol. 2000;36(4):1152-8.
Poirer NC, Pelletier LC, Pellerin M, Carrier M. 15-year experience with the Carpentier-Edwards pericardial bioprosthesis. Ann Thorac Surg. 1998;66(6 Suppl):S57-61.
Bourguignon T, Bouquiaux-Stablo AL, Candolfi P, Mirza A, Loardi C, May MA, et al. Very long-term outcomes of the Carpentier-Edwards Perimount valve in aortic position. Ann Thorac Surg. 2015;99(3):831-7.
Bourguignon T, Espitalier F, Pantaleon C, Vermes E, El-Arid JM, Loardi C, et al. Bioprosthetic mitral valve replacement in patients aged 65 years or younger: long-term outcomes with the Carpentier-Edwards PERIMOUNT pericardial valve. Eur J Cardiothorac Surg. 2018;54(2):302-9.
Chan V, Lam BK, Rubens FD, Hendry P, Masters R, Mesana TG, et al. Long-term evaluation of biological versus mechanical prosthesis use at reoperative aortic valve replacement. J Thorac Cardiovasc Surg. 2012;144(1):146-51.
Potter DD, Sundt TM, 3rd, Zehr KJ, Dearani JA, Daly RC, Mullany CJ, et al. Risk of repeat mitral valve replacement for failed mitral valve prostheses. Ann Thorac Surg. 2004;78(1):67-72; discussion 67-72.
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors who publish with this journal agree to the following conditions:
Copyright Transfer
In submitting a manuscript, the authors acknowledge that the work will become the copyrighted property of Siriraj Medical Journal upon publication.
License
Articles are licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License (CC BY-NC-ND 4.0). This license allows for the sharing of the work for non-commercial purposes with proper attribution to the authors and the journal. However, it does not permit modifications or the creation of derivative works.
Sharing and Access
Authors are encouraged to share their article on their personal or institutional websites and through other non-commercial platforms. Doing so can increase readership and citations.














