Bispecific Protein Engager-Armed T Cells (BATs): Next-Generation Platforms for Precision Cancer Immunotherapy
DOI:
https://doi.org/10.33192/smj.v78i5.281440Keywords:
Cancer immunotherapy, bispecific protein engager-armed T cells (BATs), bispecific antibodies, chimeric antigen receptor (CAR) T cells, tumor-associated antigensAbstract
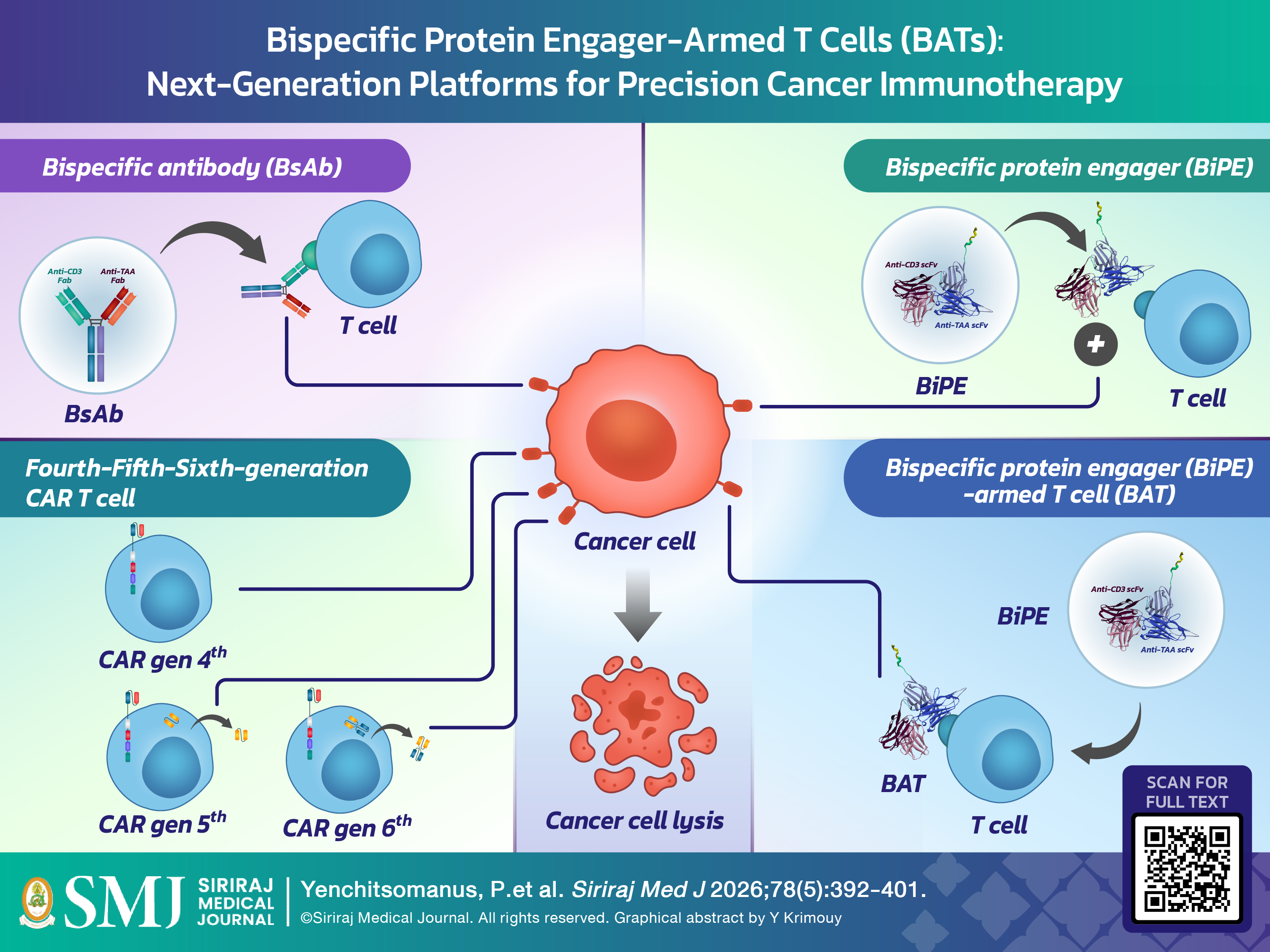
Chimeric antigen receptor (CAR) T-cell therapy and CD3-directed bispecific T-cell engagers (BiTEs) have transformed cancer immunotherapy, particularly for relapsed or refractory hematologic malignancies. However, CAR T-cell therapy remains constrained by complex genetic engineering, individualized manufacturing, specialized infrastructure, high cost, limited accessibility, and treatment-related toxicities such as cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS). In contrast, soluble BiTEs frequently require repeated dosing or continuous infusion because of their short serum half-life and may also induce CRS and neurotoxicity. To address these limitations, our research group at the Siriraj Center of Research Excellence for Cancer Immunotherapy (SiCORE-CIT) developed bispecific protein engager (BiPE)–armed T cells (BATs), a non-genetically modified and adaptable platform that combines BiPE-mediated tumor targeting with the intrinsic cytotoxic activity of activated T cells. This review positions BATs within the broader evolution of cancer immunotherapy—including monoclonal antibodies (mAbs), antibody–drug conjugates (ADCs), CAR T cells, bispecific antibodies (BsAbs), and BiTEs—and summarizes their structure and mechanism of action. BATs mediate major histocompatibility complex (MHC)-independent T-cell activation, promote immune synapse formation, and enable efficient tumor cell lysis. Clinical studies of bispecific antibody–armed T cells (BsAb-Ts), the closest clinical analogs of BATs, demonstrate feasibility, a consistent absence of dose-limiting toxicities (DLTs), and preliminary biological and clinical activity. Building on this foundation, next-generation BAT platforms targeting tumor-associated antigens and immune checkpoint molecules—including BCMA, CD138, integrin αvβ6, and PD-L1—have shown promising preclinical activity in multiple myeloma, breast cancer, and cholangiocarcinoma. Collectively, BATs represent a flexible, scalable, and potentially safer platform for next-generation precision T-cell immunotherapy.
References
Hanahan D. Hallmarks of cancer: New dimensions. Cancer Discov. 2022;12(1):31-46.
Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74(3):229-63.
International Agency for Research on Cancer. Cancer Tomorrow. Lyon: International Agency for Research on Cancer; 2024.
Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12(4):252-64.
McCarthy EF. The toxins of William B. Coley and the treatment of bone and soft-tissue sarcomas. Iowa Orthop J. 2006;26:154-8.
Hodi FS, O'Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363(8):711-23.
Rosenberg SA, Restifo NP. Adoptive cell transfer as personalized immunotherapy for human cancer. Science. 2015;348(6230):62-8.
June CH, O'Connor RS, Kawalekar OU, Ghassemi S, Milone MC. CAR T cell immunotherapy for human cancer. Science. 2018;359(6382):1361-5.
Maude SL, Frey N, Shaw PA, Aplenc R, Barrett DM, Bunin NJ, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med. 2014;371(16):1507-17.
Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med. 2017;377(26):2531-44.
Scott AM, Wolchok JD, Old LJ. Antibody therapy of cancer. Nat Rev Cancer. 2012;12(4):278-87.
Beck A, Goetsch L, Dumontet C, Corvaïa N. Strategies and challenges for the next generation of antibody-drug conjugates. Nat Rev Drug Discov. 2017;16(5):315-37.
Drago JZ, Modi S, Chandarlapaty S. Unlocking the potential of antibody-drug conjugates for cancer therapy. Nat Rev Clin Oncol. 2021;18(6):327-44.
Rafiq S, Hackett CS, Brentjens RJ. Engineering strategies to overcome the current roadblocks in CAR T cell therapy. Nat Rev Clin Oncol. 2020;17(3):147-67.
Wutti-in Y, Sujjitjoon J, Sawasdee N, Kongkhla K, Choomee K, Phanthaphol N, et al. Development of a novel anti-CD19 CAR containing a fully human scFv and three costimulatory domains. Front Oncol. 2022;11:802876.
Yuti P, Wutti-in Y, Sawasdee N, Kongkhla K, Phanthaphol N, Choomee K, et al. Anti-CD19 chimeric antigen receptor T cells secreting anti-PD-L1 single-chain variable fragment attenuate PD-L1-mediated T cell inhibition. Int Immunopharmacol. 2022;113(Pt B):109442.
Yuti P, Sawasdee N, Natungnuy K, Rujirachaivej P, Luangwattananun P, Sujjitjoon J, et al. Enhanced antitumor efficacy, proliferative capacity, and alleviation of T cell exhaustion by fifth-generation chimeric antigen receptor T cells targeting B-cell maturation antigen in multiple myeloma. Biomed Pharmacother. 2023;168:115691.
Sangsuwannukul T, Supimon K, Sujjitjoon J, Phanthaphol N, Chieochansin T, Poungvarin N, et al. Anti-tumour effect of the fourth-generation chimeric antigen receptor T cells targeting CD133 against cholangiocarcinoma cells. Int Immunopharmacol. 2020;89(Pt A):107069.
Supimon K, Sangsuwannukul T, Sujjitjoon J, Phanthaphol N, Chieochansin T, Poungvarin N, et al. Anti-mucin 1 chimeric antigen receptor T cells for adoptive T cell therapy of cholangiocarcinoma. Sci Rep. 2021;11(1):6276.
Luangwattananun P, Junking M, Sujjitjoon J, Wutti-in Y, Poungvarin N, Thuwajit C, et al. Fourth-generation chimeric antigen receptor T cells targeting folate receptor alpha antigen expressed on breast cancer cells for adoptive T cell therapy. Breast Cancer Res Treat. 2021;186(1):25-36.
Somboonpatarakun C, Phanthaphol N, Suwanchiwasiri K, Ramwarungkura B, Yuti P, Poungvarin N, et al. Cytotoxicity of fourth-generation anti-Trop2 CAR-T cells against breast cancer. Int Immunopharmacol. 2024;129:111631.
Phanthaphol N, Somboonpatarakun C, Suwanchiwasiri K, Yuti P, Chieochansin T, Junking M, et al. Enhanced cytotoxicity against cholangiocarcinoma by fifth-generation chimeric antigen receptor T cells targeting integrin αvβ6 and secreting anti-PD-L1 scFv. J Transl Med. 2025;23:308.
Wutti-In Y, Luangwattananun P, Sawasdee N, Vongchan P, Yenchitsomanus PT, Panya A. Engineering a fifth-generation CAR T cells to overcome PD-L1-mediated immunosuppression in lung cancer. Biomed Pharmacother. 2026 Feb;195:118967.
Somboonpatarakun C, Phanthaphol N, Suwanchiwasiri K, Ramwarungkura B, Yenchitsomanus PT, Junking M. Next-generation CAR T cells targeting integrin αvβ6 and PD-L1 to enhance immunotherapy for cholangiocarcinoma. Biomed Pharmacother. 2025; 193:118806.
Lee DW, Santomasso BD, Locke FL, Ghobadi A, Turtle CJ, Brudno JN, et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol Blood Marrow Transplant. 2019;25(4):625-38.
Mack M, Riethmüller G, Kufer P. A small bispecific antibody construct expressed as a functional single-chain molecule with high tumor cell cytotoxicity. Proc Natl Acad Sci U S A. 1995;92(15):7021-5.
Bargou R, Leo E, Zugmaier G, Klinger M, Goebeler M, Knop S, et al. Tumor regression in cancer patients by very low doses of a T cell-engaging antibody. Science. 2008;321(5891):974-7.
Kontermann RE. Bispecific antibodies. Drug Discov Today. 2015;20(7):838-47.
Segal DM , Weiner GJ , Weiner LM . Bispecific antibodies in cancer therapy. Curr Opin Immunol 1999;11(5):558-62.
Ridgway JBB, Presta LG, Carter P. 'Knobs-into-holes' engineering of antibody CH3 domains for heavy chain heterodimerization. Protein Eng. 1996;9(7):617-21.
Schaefer W, Regula JT, Bähner M, Schanzer J, Croasdale R, Dürr H, et al. Immunoglobulin domain crossover as a generic approach for the production of bispecific IgG antibodies. Proc Natl Acad Sci U S A. 2011;108(27):11187-92.
Carter P. Bispecific human IgG by design. J Immunol Methods. 2001;248(1-2):7-15.
Byrne H, Conroy PJ, Whisstock JC, O'Kennedy RJ. A tale of two specificities: bispecific antibodies for therapeutic and diagnostic applications. Trends Biotechnol. 2013;31(11):621-32.
Labrijn AF, Janmaat ML, Reichert JM, Parren PWHI. Bispecific antibodies: a mechanistic review of the pipeline. Nat Rev Drug Discov. 2019;18(8):585-608.
Yankelevich M, Thakur A, Modak S, Chu R, Taub J, Martin A, et al. Targeting refractory/recurrent neuroblastoma and osteosarcoma with anti-CD3×anti-GD2 bispecific antibody armed T cells. J Immunother Cancer. 2024;12(3):e008744.
Fadul CE, Thakur A, Kim J, Kassay-McAllister J, Schalk D, Lopes MB, et al. Phase I study targeting newly diagnosed grade 4 astrocytoma with bispecific antibody armed T cells (EGFR BATs) in combination with radiation and temozolomide. J Neurooncol. 2024;166(2):321-30.
Lum LG, Al-Kadhimi Z, Deol A, Kondadasula V, Schalk D, Tomaszewski E, et al. Phase II clinical trial using anti-CD3 × anti-HER2 bispecific antibody armed activated T cells (HER2 BATs) consolidation therapy for HER2-negative (0-2+) metastatic breast cancer. J Immunother Cancer. 2021;9(6):e002194.
Vaishampayan UN, Thakur A, Chen W, Deol A, Patel M, Dobson K, et al. Phase II trial of pembrolizumab and anti-CD3 × anti-HER2 bispecific antibody armed activated T cells in metastatic castrate-resistant prostate cancer. Clin Cancer Res. 2023;29(1):122-32.
Lum LG, Thakur A, Choi M, Deol A, Kondadasula V, Schalk D, et al. Clinical and immune responses to anti-CD3 × anti-EGFR bispecific antibody armed activated T cells (EGFR BATs) in pancreatic cancer patients. Oncoimmunology. 2020;9(1):1773201.
Lum LG, Thakur A, Kondadasula SV, Al-Kadhimi Z, Deol A, Tomaszewski EN, et al. Targeting CD138-/CD20+ Clonogenic Myeloma Precursor Cells Decreases These Cells and Induces Transferable Antimyeloma Immunity. Biol Blood Marrow Transplant. 2016;22(5):869-78.
Goebeler ME, Bargou RC. Blinatumomab: a CD19/CD3 bispecific T cell engager (BiTE) with unique anti-tumor efficacy. Cancer Treat Rev. 2016;49:63-76.
Kantarjian H, Stein A, Gökbuget N, Fielding AK, Schuh AC, Ribera JM, et al. Blinatumomab versus chemotherapy for advanced acute lymphoblastic leukemia. N Engl J Med. 2017;376(9):836-47.
Jen EY, Xu Q, Schetter A, Przepiorka D, Shen YL, Roscoe D, et al. FDA approval: blinatumomab for patients with B-cell precursor acute lymphoblastic leukemia in morphologic remission with minimal residual disease. Clin Cancer Res. 2019;25(2):473-7.
U.S. Food and Drug Administration. FDA granted accelerated approval to blinatumomab (Blincyto, Amgen Inc.) for the treatment of adult and pediatric patients with B-cell precursor acute lymphoblastic leukemia. U.S. Food and Drug Administration; 2018.
Moreau P, Garfall AL, van de Donk NWCJ, Nahi H, San-Miguel J, Oriol A, et al. Teclistamab in relapsed or refractory multiple myeloma. N Engl J Med. 2022;387(6):495-505.
Lesokhin AM, Tomasson MH, Arnulf B, Mikhael JR, Minnema MC, Krishnan A, et al. Elranatamab in patients with relapsed or refractory multiple myeloma. Nat Med. 2023;29(9):2259-67.
Nathan P, Hassel JC, Rutkowski P, Baurain JF, Butler MO, Schlaak M, et al. Overall survival benefit with tebentafusp in metastatic uveal melanoma. N Engl J Med. 2021;385(13):1196-206.
Hassel JC, Piperno-Neumann S, Schankin C, Rutkowski P, Carvajal RD, Butler MO, et al. Three-year overall survival with tebentafusp in metastatic uveal melanoma. N Engl J Med. 2023;389(24):2256-66.
Luangwattananun P, Sangsuwannukul T, Supimon K, Thuwajit C, Chieochansin T, Sa-nguanraksa D, et al. Anti-PD-L1 × anti-CD3 bispecific T-cell engager-armed T cells can overcome immunosuppression and redirect T cells to kill breast cancer cells expressing PD-L1. Int Immunopharmacol. 2023;124:111012.
Supimon K, Sangsuwannukul T, Luangwattananun P, Yenchitsomanus PT. Enhanced cytotoxicity in multiple myeloma via T cells armed with bispecific T cell engager targeting B-cell maturation antigen on cancer cells and CD3 on T cells. Int Immunopharmacol. 2024;143(Pt 2):113480.
Songprakhon P, Luangwattananun P, Choomee K, Sawasdee N, Somboonpatarakun C, Chieochansin T, et al. CD138-targeted bispecific protein engager-armed T cells exhibit potent and selective cytotoxicity against multiple myeloma cells. Int Immunopharmacol. 2026;174:116295.
Suwanchiwasiri K, Somboonpatarakun C, Phanthaphol N, Yuti P, Chieochansin T, Junking M, et al. Bispecific T cell engager-armed T cells targeting integrin αvβ6 exhibit enhanced T cell redirection and antitumor activity in cholangiocarcinoma. Biomed Pharmacother. 2024;175:116718.
Dustin ML, Long EO. Cytotoxic immunological synapses. Immunol Rev. 2010;235(1):24-34.
Stinchcombe JC, Griffiths GM. Secretory mechanisms in cell-mediated cytotoxicity. Annu Rev Cell Dev Biol. 2007;23:495-517.
Huse M. Mechanical forces in the immune system. Nat Rev Immunol. 2017;17(11):679-90.
Dustin ML. The immunological synapse. Cancer Immunol Res. 2014;2(11):1023-33.
Voskoboinik I, Whisstock JC, Trapani JA. Perforin and granzymes: function, dysfunction and human pathology. Nat Rev Immunol. 2015;15(6):388-400.
Wathikthinnakon M, Sittithumcharee G, Chen S, Prasopporn S, Choomee K, Chieochansin T, et al. Gemcitabine and anti-PD-L1 × anti-CD3 bispecific T-cell engager synergistically enhance T-cell cytotoxicity against cholangiocarcinoma cells. Sci Rep. 2022;12(1):6154.
Sawasdee N, Songprakhon P, Luangwattananun P, Rujirachaivej P, Yuti P, Yenchitsomanus PT. Low-dose bortezomib enhances the cytotoxicity of anti-PD-L1 × anti-CD3 bispecific engager-armed T cells against multiple myeloma cells. Biomed Pharmacother. 2025;184:117878.
Wherry EJ, Kurachi M. Molecular and cellular insights into T cell exhaustion. Nat Rev Immunol. 2015;15(8):486-99.
Depil S, Duchateau P, Grupp SA, Mufti G, Poirot L. 'Off-the-shelf' allogeneic CAR T cells: development and challenges. Nat Rev Drug Discov. 2020;19(3):185-99.
Fesnak AD, June CH, Levine BL. Engineered T cells: the promise and challenges of cancer immunotherapy. Nat Rev Cancer. 2016;16(9):566-81.
Vivier E, Tomasello E, Baratin M, Walzer T, Ugolini S. Functions of natural killer cells. Nat Immunol. 2008;9(5):503-10.
Mackensen A, Müller F, Mougiakakos D, Böltz S, Wilhelm A, Aigner M, et al. Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus. Nat Med. 2022;28(10):2124-32.
Vu T, Gwathmey K, Benatar M, Cutter G, Claussen G, Burns TM, et al. BCMA-directed mRNA CAR-T cell therapy for myasthenia gravis: a randomized, double-blind, placebo-controlled phase 2b trial. Nat Med. 2026 Jan 9. doi: 10.1038/s41591-025-04171-y.
Published
How to Cite
License
Copyright (c) 2026 Siriraj Medical Journal

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors who publish with this journal agree to the following conditions:
Copyright Transfer
In submitting a manuscript, the authors acknowledge that the work will become the copyrighted property of Siriraj Medical Journal upon publication.
License
Articles are licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License (CC BY-NC-ND 4.0). This license allows for the sharing of the work for non-commercial purposes with proper attribution to the authors and the journal. However, it does not permit modifications or the creation of derivative works.
Sharing and Access
Authors are encouraged to share their article on their personal or institutional websites and through other non-commercial platforms. Doing so can increase readership and citations.














